Do you know where the metals are located on the periodic table?
If not, you’ve come to the right place.
Let’s answer where the metals can be found on the periodic table.
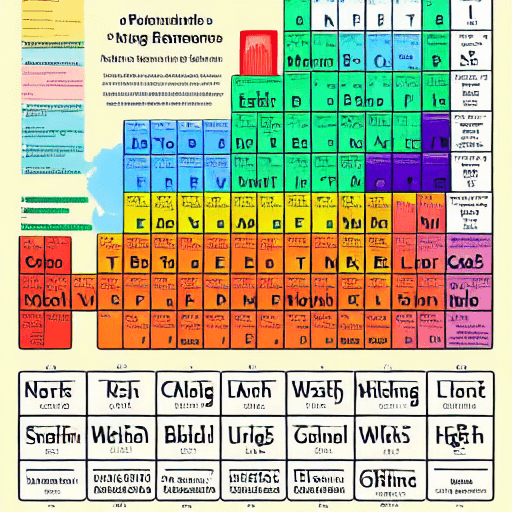
Metals are primarily found on the left side and in the middle of the periodic table.
Here is an overview of where metals are located:
- Alkali metals – Group 1 (IA) – e.g. lithium, sodium, potassium
- Alkaline earth metals – Group 2 (IIA) – e.g. beryllium, magnesium, calcium
- Transition metals – Groups 3-12 (IIIB – IIB) – e.g. iron, copper, zinc, silver
- Basic metals – Groups 13-15 (IIIA – VA) – e.g. aluminum, tin, lead
- Lanthanides – Inner transition metals in row 6
- Actinides – Inner transition metals in row 7
- Metallic elements – Right side of the table along the stair-step line from boron to polonium
- Metalloids – Along zig-zag line from boron to astatine – have intermediate properties
- Nonmetals – Upper right corner (e.g. nitrogen, oxygen) and the halogens
So in summary, the metals occupy much of the table, with the most reactive types on the left and the most noble metals on the right.
The upper right has primarily nonmetals. The metalloids form a diagonal separating the metals and nonmetals.
Metals are located in the left column of the periodic table. This column is called the “metals” group.
Elements in this group are characterized by their metallic properties, which include a lustrous appearance, good electrical conductivity, and the ability to form alloys with other metals.
Where are metals located on the periodic table?
Metals are located on the left side of the periodic table, and they are characterized by their ability to conduct electricity and heat.
Explain It To A Child
The left column of the periodic table is called the “metals” group. Elements in this group are characterized by their metallic properties. Metallic properties include a lustrous appearance, good electrical conductivity, and the ability to form alloys with other metals.
They also have a lustrous appearance and are malleable, meaning that they can be hammered into thin sheets. The most abundant metals in the Earth’s crust include iron, aluminum, copper, and lead.
These elements play an important role in our everyday lives, and they can be found in everything from buildings and bridges to coins and jewelry.
Which group in the periodic table contains metals?
How to identify METALS NONMETALS and METALLOIDS on the PERIODIC TABLE
When it comes to the periodic table, there are a few things that you need to know in order to understand which group contains the metals.

- First, remember that the groups are arranged according to increasing atomic number.
- So, the first group will have elements with low atomic numbers, while the last group will have elements with high atomic numbers.
- Secondly, remember that metals are found on the left side of the table, while non-metals are found on the right side.
- With these two things in mind, you should be able to determine which group contains the metals.
As you can see, group 1 contains metals such as lithium and sodium, while group 2 contains metals such as magnesium and calcium.
So, when it comes to the periodic table, metals are definitely not limited to just one group!
Why are metals on the left of the periodic table?

The periodic table is arranged in order of increasing atomic number. Metals are elements that have a tendency to lose electrons, giving them a positive charge.
The further to the left on the periodic table an element is, the less inclination it has to lose electrons.
So, elements on the left side of the periodic table are more likely to be metals than those on the right side.
There are exceptions to this trend, such as hydrogen, which is a nonmetal despite being on the left side of the periodic table.
However, in general, metals are found on the left side of the table because they have a greater tendency to lose electrons than elements on the right side.
Where are the most reactive metals on the periodic table found?
The most reactive metals are found in the far left column of the periodic table, which is known as the alkali metal family.
These metal elements include:
- lithium
- sodium
- potassium
- rubidium
- cesium
- francium
Alkali metals are extremely reactive, and they are known for their explosive reaction with water. In fact, caution must be taken when handling these materials, as they can cause serious injury if not handled properly.
When exposed to air, alkali metals oxidize quickly, forming a white powder on their surface. This process is called corrosion, and it can cause the metal to break down over time.
For this reason, alkali metals are often used in industries where corrosion resistance is important, such as in the construction of power plants and chemical plants.
Where are the transition metals on the periodic table?
At the center of the periodic table are the transition metals, so named because they occupy the transition between the s-block and d-block elements.
Transition metals are characterized by their high melting and boiling points, as well as their ductility and malleability. They are also good conductors of heat and electricity.
In addition to their practical applications, transition metals are also responsible for the beauty of many objects in our world, from the lustrous sheen of gold to the brilliant colors of copper sulfate crystals.
Whether we realize it or not, transition metals play an important role in our lives.
Where are alkali earth metals on the periodic table
The alkali earth metals are located in Group 2 of the periodic table. This group includes elements such as magnesium, calcium, and strontium.
These elements are all characterized by their high reactivity.
- They have low melting points and boiling points, and they readily form oxides and hydroxides. Alkali earth metals are found in many everyday objects, such as eggshells, antacid tablets, and fire extinguishers.
- They are also used in a variety of industrial applications, such as metallurgy and chemical production.
- Although they are essential to many different fields, alkali earth metals can be dangerous if not handled properly.
They are highly reactive, so they must be stored carefully to avoid accidents.
